Dr. Marian Olaru, Dr. Stefan Mebs, Prof. Dr. Jens Beckmann
Angewandte Chemie International Edition (2021) 60, 19133-19138
https://doi.org/10.1002/anie.202107975
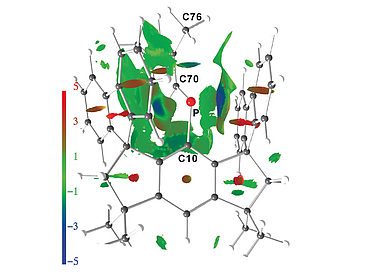
Carbenes and their analogues have constantly enthralled chemists with their intriguing reactivity of ambiphilic character stemming from their electronic structures. Phosphenium and arsenium ions are fiercely reactive cationic species, the stabilization of which has been so far achieved in the condensed phase by dispersing the positive charge through electromeric conjugation with at least one electron-rich substituent (frequently amido groups). Although observed in the gas phase, the isolation of dicoordinate phosphenium and arsenium ions lacking such stabilizing ligands has eluded chemists for decades. Herein we show that by judicious choice of aromatic substituents, dicoordinate, donor-free, Lewis-superacidic phosphenium and arsenium ions can be kinetically stabilized. They feature singlet electronic ground states possessing a vacant p-orbital and an electron lone pair with predominantly s-character.
© The Authors, https://doi.org/10.1002/anie.202107975, licensed under CC BY 4.0